As remediation practitioners we continually seek to improve the effectiveness of remediation processes in order to bring greater value to our clients. Increasing expectations for more holistic remedies encourage advancements in in-situ remediation chemistry to incorporate multiple, synergistic processes in order to keep pace. This blog describes one example of an innovation to enhance performance of zero valent iron (ZVI) technology, which has been widely used for chlorinated solvent remediation for over three decades. The innovation is to incorporate vitamin B12 to facilitate more effective contaminant degradation and expand the scope of contaminants that can be treated by ZVI.
THE PROBLEM – MINIMAL REACTIVITY OF SOME CHLORINATED SOLVENTS WITH ZVI
Certain types of chlorinated solvents, called chlorinated alkanes, are relatively unreactive in the subsurface to common in-situ remedial approaches. These compounds include 1,2-dichloroethane (1,2 DCA), 1,2,3-trichloropropane (1,2,3 TCP), and 1,2-dichloropropane (1,2-DCP). Destruction of these compounds requires higher amounts of energy to break their chemical bonds than is normally provided by ZVI and other types of reagents utilized for in-situ remediation. These compounds are also considered emerging contaminants of concern, with growing focus from regulatory agencies. Thus, we undertook research to evaluate mechanisms to improve readily deployable in-situ remedial approaches such as ZVI.
THE IDEA – CATALYZED ZVI FOR ABIOTIC DESTRUCTION OF CHLORINATED ALKANES
Vitamin B12, also known as cobalamin, is a naturally occurring compound. It is known as an organometallic compound, meaning there is at least one chemical bond between a carbon atom of an organic molecule and a metal. Vitamin B12, which has a cobalt atom situated at the center of its structure, acts as a redox mediator that shuttles electrons during reactions due to the multiple oxidation states of the cobalt. Vitamin B12 is also known to assist in biological processes that include anaerobic bioremediation and has also been shown to facilitate the abiotic reduction of chlorinated ethene compounds in the presence of strong reductants. The high water solubility and nontoxic characteristics of vitamin B12 make it a great candidate to evaluate as a catalyst for catalyzing reactions between ZVI and 1,2 DCA, 1,2 DCP and 1,2,3 TCP, which can enhance in-situ degradation of these compounds.
TESTING THE IDEA – LABORATORY STUDIES CONFIRM COMPLETE ABIOTIC DESTRUCTION
The first step was to determine if ZVI could chemically reduce vitamin B12 to B12r , also called reduced B12. This is important because B12r has an unpaired electron; this allows B12r to act as a redox mediator and enhance the reactivity of ZVI towards the chlorinated alkanes. The conversion of B12 to B12r was confirmed by characterizing how the absorbance of light in water solutions containing vitamin B12 and ZVI changed.
The next step was to evaluate the reactivity of chlorinated alkanes to ZVI amended with vitamin B12. Experiments demonstrated that solutions amended with ZVI and vitamin B12 can catalyze the degradation of all chlorinated alkanes tested, resulting in complete destruction. Destruction was confirmed by observations of predicted byproducts and increase concentration of free chloride. Also, redox cycling between B12 – B12r indicated that the catalytic effect of the B12 will be sustained for an extended period of time. Similar experiments performed in the absence of vitamin B12, in contrast, exhibited no significant degradation (Figure 1).
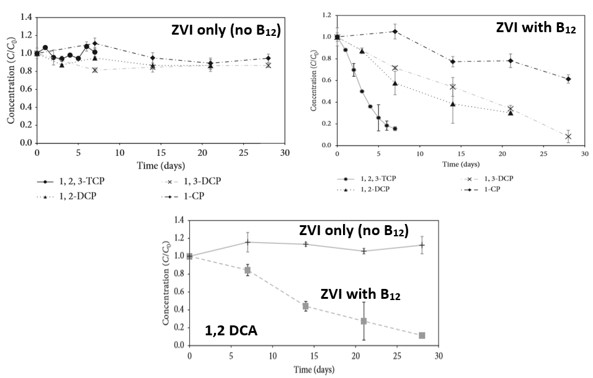
Figure 1
N. Lapeyrouse, M. Liu, S. Zou, G. Booth and C. Yestrebsky, “Remediation of chlorinated alkanes by vitamin B12 and zero-valent iron,” Journal of Chemistry, vol. 2019, Article ID 7565464, 8 pages
PRACTICAL APPLICATION OF NEW SOLUTION – INCORPORATED INTO EZVI TECHNOLOGY
Emulsified zero valent iron (EZVI) products are already utilized for in-situ contaminant destruction. An EZVI formulation was prepared with vitamin B12 to evaluate if this approach may be effective for direct destruction of chlorinated alkane dense non-aqueous phase liquids (DNAPLs). Lab tests demonstrated that degradation byproducts were observed in the B12 amended emulsions, while no byproducts were observed for the unamended emulsions, indicating that the B12 catalyst could be integrated into the EZVI technology. The B12 was added into the interior of the emulsion, which contains ZVI powder suspended within a water drop and wrapped within a vegetable oil membrane. You can think of these as miniature reactors that can be injected directly into the most contaminated areas of a remediation site. The structure of the EZVI emulsion enables the B12 to be added at an appropriate concentration with the ZVI and protects it from direct contact with groundwater, which would dilute the water-soluble B12. The chlorinated alkane DNAPLs absorb into the vegetable oil membrane and then diffuse into the water drop interior of the emulsion, where they are destroyed by contact with the B12 amended ZVI (Figure 2). This development has potentially created a new injectable alternative to address chlorinated alkane source mass for our clients and highlights how Woodard & Curran can innovate with purpose to address client concerns.

Figure 2
